Anomeric carbon in fructose10/10/2023 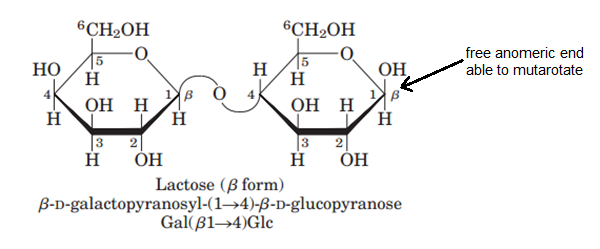
If it is directed in the opposite direction it is the β-anomer. Which carbon is the anomeric carbon A) 1 B) 2 OH 1 c) 3 2 OH D) 4 E) 5 3 I Subm 6 HOH C 5 4 OH The Haworth projection of D-fructose is shown. Question: 6 5 4 The Haworth projection of D-altrose is shown. Are you looking for an answer to the topic Where are the anomeric carbons in lactose We answer all your questions at the website in category: +15 Marketing Blog Post Ideas And Topics For You.You will find the answer right below. 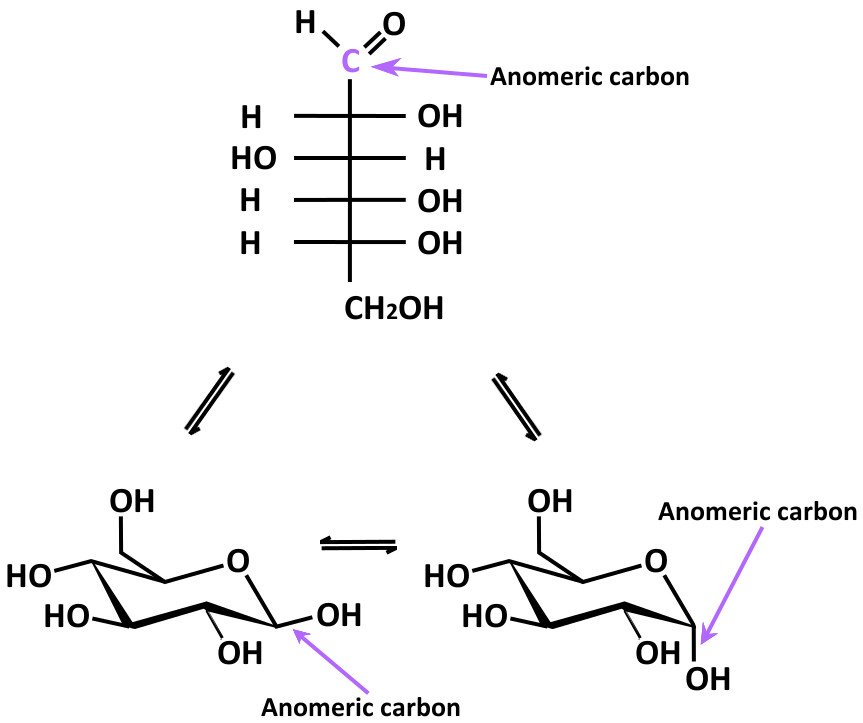
In a Fischer projection, if the substituent off the anomeric centre is on the same side as the oxygen of the configurational (D- or L-) carbon, then it is the α-anomer. This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. The configuration at the anomeric centre (that derived from the carbonyl carbon) is denoted alpha- (α-) or beta- (β-) by reference to the stereocentre that determines the absolute configuration. The anomeric carbon atoms 2 and 1 are chiral, and two configurations are possible for the OH group. Hemiacetal formation between carbons 6 and 1 leads to the pyranose structure. The deoxy- designation refers to the lack of an alcohol, -OH, group as will be shown in detail further down. Ribose is used in R NA and deoxyribose is used in D NA. They are diastereoisomers of one another. Cyclic hemiacetal (hemiketal) formation between carbon 5 and carbon 2 of fructose leads to the furanose structure. Ribose and its related compound, deoxyribose, are the building blocks of the backbone chains in nucleic acids, better known as DNA and RNA. The two stereoisomers formed from the two possible stereochemistries at the anomeric centre are called anomers. The anomeric centre of a sugar is a stereocentre created from the intramolecular formation of an acetal (or ketal) of a sugar hydroxyl group and an aldehyde (or ketone) group. If you would like to suggest an addition or correction, please contact the page's Responsible Curator directly by e-mail. Although we have been looking at specific examples for glucose and fructose, other five- and six-carbon monosaccharides also exist in solution as equilibrium mixtures of open chains and cyclic intramolecular hemiacetals and hemiketals. These two cyclic forms differ only in the configuration of the hydroxyl group, called anomeric carbon. CAZypedia is a living document, so further improvement of this page is still possible. This carbon is called the anomeric carbon in carbohydrate terminology. Identify anomeric carbon atoms in monosaccharide units. This page has been approved by the Responsible Curator as essentially complete. Structure of disaccharide formed by glucose and fructose is given below.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
